
1)Fill in the blanks.
(a) Most liquids that conduct electricity are solutions of
__acids__, __bases__ and __salts__.
__acids__, __bases__ and __salts__.
(b) The passage of an electric current through a solution causes __chemical__
effects.
effects.
(c) If you pass current through copper sulphate solution, copper
gets deposited on the plate connected
to the __negative__ terminal of the battery.
gets deposited on the plate connected
to the __negative__ terminal of the battery.
(d) The process of depositing a layer of any desired metal on
another material by means of electricity
is called _electroplating__.
another material by means of electricity
is called _electroplating__.
2)When the free ends of a tester are dipped into a solution, the
magnetic needle shows deflection. Can you explain the reason?
magnetic needle shows deflection. Can you explain the reason?
ANSWER:-The deflection in the compass needle shows that current is
flowing through the wounded
wire and hence, through the circuit. The circuit is complete since free ends of
the tester are dipped in a solution. The solution is certainly a conducting solution. This is the reason why the compass
needle shows a deflection.
flowing through the wounded
wire and hence, through the circuit. The circuit is complete since free ends of
the tester are dipped in a solution. The solution is certainly a conducting solution. This is the reason why the compass
needle shows a deflection.
3)Name three liquids, which when tested in the manner shown in
Fig. 14.9, may cause the magnetic needle to deflect.(diagram refr to the book)
Fig. 14.9, may cause the magnetic needle to deflect.(diagram refr to the book)
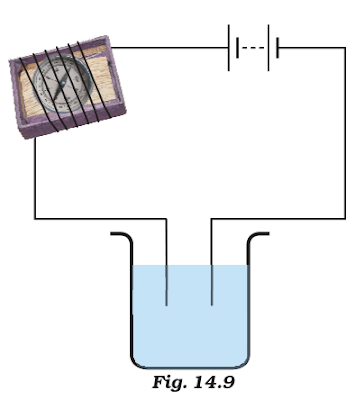
ANSWER:- Liquids like lemon juice, salt water and vegetable oil allow
electricity to pass through them. Hence, these liquids can be used as in the
beaker to show the given effect.
electricity to pass through them. Hence, these liquids can be used as in the
beaker to show the given effect.
4) The bulb does not glow in the setup shown in Fig. 14.10. List
the possible reasons. Explain your answer. (diagram refr to the book)
the possible reasons. Explain your answer. (diagram refr to the book)
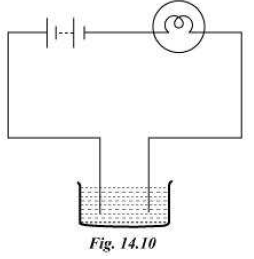
ANSWER:- The bulb may not glow because of the following reasons:
(i) Liquid in the beaker is non-conducting. In such case, the
electric current would not be able to pass through the liquid. Hence, the circuit is not
complete.
electric current would not be able to pass through the liquid. Hence, the circuit is not
complete.
(ii) Electric current in the circuit is very weak. This can happen
if the material used for making the circuit is not a good conductor of
electricity or the battery does not have sufficient energy to generate
electricity.
if the material used for making the circuit is not a good conductor of
electricity or the battery does not have sufficient energy to generate
electricity.
5)A tester is used to check the conduction of electricity
through two liquids, labelled A and B.
through two liquids, labelled A and B.
It is found that the bulb of the tester glows brightly for
liquid A while it glows very dimly for liquid B. You would conclude that
liquid A while it glows very dimly for liquid B. You would conclude that
(i) liquid A is a better conductor than liquid B.
(ii) liquid B is a better conductor than liquid A.
(iii) both liquids are equally conducting.
(iv) conducting properties of liquid cannot be compared in this
manner.
manner.
ANSWER:- (i) Liquid A is a better conductor than liquid B. The amount of
current flowing through a conducting solution depends on the conductivity of
the solution. With more conductivity, more current passes through the solution
and vice-versa.
current flowing through a conducting solution depends on the conductivity of
the solution. With more conductivity, more current passes through the solution
and vice-versa.
6)Does pure water conduct electricity? If not, what can we do to
make it conducting?
make it conducting?
ANSWER:- No. Pure water does not conduct electricity.
Pure water can conduct electricity when a pinch of
common salt is added to it, as salt solution is conducting
in nature.
Pure water can conduct electricity when a pinch of
common salt is added to it, as salt solution is conducting
in nature.
7)In case of a fire, before the firemen use the water hoses,
they shut off the main electrical supply for the area. Explain why they do
this.
they shut off the main electrical supply for the area. Explain why they do
this.
ANSWER:- Water may conduct electricity. If the electrical supply for the
area is not shut off and water is poured over electrical appliances, then
electricity may pass through water and harm the firemen. That is why, in case
of a fire, the firemen shut off the main electrical supply for the area before
they use the water hoses.
area is not shut off and water is poured over electrical appliances, then
electricity may pass through water and harm the firemen. That is why, in case
of a fire, the firemen shut off the main electrical supply for the area before
they use the water hoses.
8)A child staying in a coastal region tests the drinking water
and also the seawater with his tester. He finds that the compass needle
deflects more in the case of seawater. Can you explain the reason?
and also the seawater with his tester. He finds that the compass needle
deflects more in the case of seawater. Can you explain the reason?
ANSWER:- Sea water contains more dissolved salts than the
drinking water. Hence, it is more conducting than the
drinking water. Because of this reason, the compass
needle deflects more
drinking water. Hence, it is more conducting than the
drinking water. Because of this reason, the compass
needle deflects more
9)Is it safe for the electrician to carry out electrical repairs
outdoors during heavy downpour? Explain.
outdoors during heavy downpour? Explain.
ANSWER:- No. It is not safe to repair electrical appliances
outdoors during heavy downpour. This is because rain
water contains dissolved salts. Therefore, rain water can conduct electricity.
outdoors during heavy downpour. This is because rain
water contains dissolved salts. Therefore, rain water can conduct electricity.
10)Paheli had heard that rain water is as good as distilled
water. So, she collected some rain water in a clean glass tumbler and tested it
using a tester. To her surprise, she found that the compass needle showed
deflection. What could be the reasons?
water. So, she collected some rain water in a clean glass tumbler and tested it
using a tester. To her surprise, she found that the compass needle showed
deflection. What could be the reasons?
ANSWER:-Rain water contains dissolved salts. This makes it a conducting
solution. There are no dissolved salts present in the distilled water. Hence,
rain water can allow electricity to pass through it while distilled water
cannot.
solution. There are no dissolved salts present in the distilled water. Hence,
rain water can allow electricity to pass through it while distilled water
cannot.
11) Prepare a list of
objects around you that are electroplated.
objects around you that are electroplated.
ANSWER:- (i) Chromium plating is done on different parts of cars, buses
and motor cycles to give them shiny appearance.
and motor cycles to give them shiny appearance.
ii)Iron used in constructing a building is coated with a layer of
zinc. This protects iron from corrosion
and rusting.
zinc. This protects iron from corrosion
and rusting.
iii)A fine layer of gold is deposited on the silver ornaments and
they are called gold-plated ornaments.
they are called gold-plated ornaments.
12) The process that you saw in Activity 14.7 is used for
purification of copper. A thin plate of pure copper and a thick rod of impure
copper are used as electrodes. Copper from impure rod is sought to be
transferred to the thin copper plate. Which electrode should be attached to the
positive terminal of the battery and why?
purification of copper. A thin plate of pure copper and a thick rod of impure
copper are used as electrodes. Copper from impure rod is sought to be
transferred to the thin copper plate. Which electrode should be attached to the
positive terminal of the battery and why?
ANSWER:- Copper ion is positively charged. It is attracted towards the
plate which is connected to the negative terminal of the battery. As copper
ions are transferred to the thin copper plate, this thin pure copper plate must
be connected to the negative
terminal of the battery. Consequently, impure copper rod is connected to the
positive terminal.
plate which is connected to the negative terminal of the battery. As copper
ions are transferred to the thin copper plate, this thin pure copper plate must
be connected to the negative
terminal of the battery. Consequently, impure copper rod is connected to the
positive terminal.