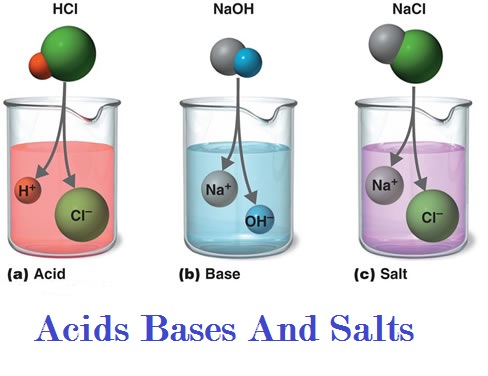
1) State differences between acids and
bases?
bases?
ANSWER:-
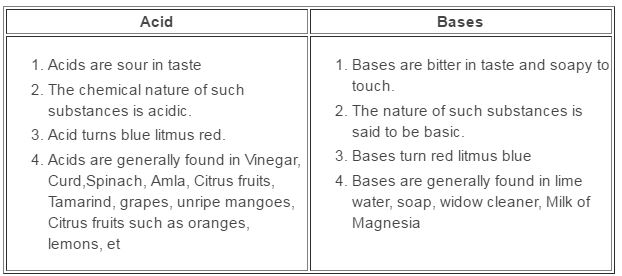
2) Ammonia is found in many household products, such as window
cleaners. It turns red litmus blue. What is its nature?
cleaners. It turns red litmus blue. What is its nature?
ANSWER:-Ammonia is basic in nature as it turns the colour of red litmus
paper to blue.
paper to blue.
3) Name the source from which litmus solution is obtained. What
is the use of this solution?
is the use of this solution?
ANSWER:-Litmus solution is obtained from lichens. A natural dye extracted
from lichens is dissolved in distilled water to obtain litmus solution. It is used
as an indicator to distinguish between acids and bases.
from lichens is dissolved in distilled water to obtain litmus solution. It is used
as an indicator to distinguish between acids and bases.
4) Is the distilled water acidic/basic/ neutral? How would you
verify it?
verify it?
ANSWER: – Distilled water is neutral in nature. The same can be verified by
using red and blue litmus papers.
Neither will show a colour change with distilled water. This proves that distilled water is neutral.
using red and blue litmus papers.
Neither will show a colour change with distilled water. This proves that distilled water is neutral.
5) Describe the process of neutralization
with the help of an example.
with the help of an example.
ANSWER: – In
this reaction, both acid and base cancel each other’s effect. Neutralisation reaction results in the
formation of salt and water. During this reaction,
energy in the form of heat is evolved.
this reaction, both acid and base cancel each other’s effect. Neutralisation reaction results in the
formation of salt and water. During this reaction,
energy in the form of heat is evolved.
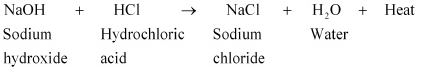
Acid + Base → Salt + Water + Heat
For example, when sodium hydroxide (NaOH) is added to hydrochloric
acid (HCl), sodium chloride (NaCl) and water (H2O) are obtained.
acid (HCl), sodium chloride (NaCl) and water (H2O) are obtained.
6) Mark ‘T’ if the statement is true
and ‘F’ if it is false:
and ‘F’ if it is false:
(i) Nitric acid turns red
litmus blue. (F)
litmus blue. (F)
(ii) Sodium hydroxide turns blue litmus red. (F)
(iii) Sodium hydroxide and hydrochloric acid neutralise each other
and form salt and water. (T)
and form salt and water. (T)
(iv) Indicator is a substance which shows different colours in
acidic and basic solutions. (T)
acidic and basic solutions. (T)
(v) Tooth decay is caused by the presence of a base. (F)
8) Explain why:
(a) An antacid tablet is taken when
you suffer from acidity.
you suffer from acidity.
(b) Calamine solution is applied on
the skin when an ant bites.
the skin when an ant bites.
(c) Factory waste is neutralised
before disposing it into the water bodies.
before disposing it into the water bodies.
ANSWER :-(a) This is because during acidity, an excess of acids is produced
in the stomach. An antacid
contains base, such as milk of magnesia. These bases react with excess of acids
and neutralize their effect, thus giving us relief.
in the stomach. An antacid
contains base, such as milk of magnesia. These bases react with excess of acids
and neutralize their effect, thus giving us relief.
(b) When an ant bites, it injects formic acid into the skin.
Calamine solution contains zinc
carbonate which is basic in nature. Therefore, it is applied on the skin to
neutralize the effect of formic acid.
Calamine solution contains zinc
carbonate which is basic in nature. Therefore, it is applied on the skin to
neutralize the effect of formic acid.
(c) Factory wastes contain acids. Therefore, these wastes, when
thrown directly to water bodies, harm aquatic lives. Hence, these
wastes are neutralised with basic chemicals before disposing to water bodies.
thrown directly to water bodies, harm aquatic lives. Hence, these
wastes are neutralised with basic chemicals before disposing to water bodies.
9) Three liquids are given to you. One is hydrochloric acid,
another is sodium hydroxide and the third is a sugar solution. How will you identify
them? You have only turmeric indicator.
another is sodium hydroxide and the third is a sugar solution. How will you identify
them? You have only turmeric indicator.
ANSWER:-The substances Effect on turmeric indicator
1. Hydrochloric acid Yellow to blue
2. Sodium hydroxide Yellow to red
3. Sugar solution No change
10) Blue litmus paper is dipped in a solution. It remains blue.
What is the nature of the solution? Explain.
What is the nature of the solution? Explain.

ANSWER :(i) It can be identified on the basis of the following
observations : Bases change the colour
of litmus paper to blue. As the colour of blue litmus paper is not affected,
the solution must be basic.
observations : Bases change the colour
of litmus paper to blue. As the colour of blue litmus paper is not affected,
the solution must be basic.
(ii) If the solution is neutral, even then colour of litmus will
not change.
not change.
11) Consider the following statements:
(a) Both acids and bases change colour of all indicators.
(b) If an indicator gives a colour change with an acid, it does
not give a change with a base.
not give a change with a base.
(c) If an indicator changes colour with a base, it does not
change colour with an acid.
change colour with an acid.
(d) Change of colour in an acid and a base depends on the type
of the indicator. Which of these statements are correct?
of the indicator. Which of these statements are correct?
(i) All four (ii) (a) and (d) (iii) (b) and (c) (iv) only (d)
ANSWER:-(ii) (a) and (d)