1)
What are macromolecules?
Give Example.
What are macromolecules?
Give Example.
Macromolecules
are large sized chemicals which get separated in acid insoluble fraction using
trichloro acetic acid. They are polymers with a molecular mass of 1000 Daltons or
above e.g. = Proteins, lipids.
are large sized chemicals which get separated in acid insoluble fraction using
trichloro acetic acid. They are polymers with a molecular mass of 1000 Daltons or
above e.g. = Proteins, lipids.
2. Illustrate a glycoside,
peptide and phosphodiester bond.
peptide and phosphodiester bond.
Glycosidic bond: It is a bond formed during dehydrate
synthesis of polysaccharide with one monosaccharide providing hydroxyl (-OH)
and other providing hydrogen ((-H) to form a molecules of water (HOH)
synthesis of polysaccharide with one monosaccharide providing hydroxyl (-OH)
and other providing hydrogen ((-H) to form a molecules of water (HOH)
The bond is generally –COC – or – CNC – C5H10O5
x HCOH + Ox CHO5H10C5
x HCOH + Ox CHO5H10C5
C5H10O5xHCOCHxO5H10C5+HO
Peptide
bond: It is formed during
dehydrate synthesis of a peptide with one amino acid and hydrogen to form water
bond: It is formed during
dehydrate synthesis of a peptide with one amino acid and hydrogen to form water
HNxN – CH – COOH
– ChxCoxNH – CHxCOOH + H2o
H2NxCHxCOOH +
HNxN – CH – COOH
– ChxCoxNH – CHxCOOH + H2o
HNxN – CH – COOH
– ChxCoxNH – CHxCOOH + H2o
R R R R
Phosphodiester
Bond:
is a double ester bond formed during dehydrate synthesis in the
Bond:
is a double ester bond formed during dehydrate synthesis in the
Reaction between a phosphoric acid (HOHPO2OH) and two
Pentose sugar. The bond is –OXHPO2x O- C4H7O3
x CH2OH +
HOHPO2 x OH + HOCH2O3H7C4
Pentose sugar. The bond is –OXHPO2x O- C4H7O3
x CH2OH +
HOHPO2 x OH + HOCH2O3H7C4
C4H7O3.CH2O x HPO2OCH2O3H7O4
+ 2H2O
+ 2H2O
3. What do you meant by tertiary structure of
proteins?
proteins?
It is a structure formed by folding of
secondarily coiled polypeptide,a-helix and B
pleated in such a way as to bring the functional side groups to the surface and
inactive side group in the interior.
secondarily coiled polypeptide,a-helix and B
pleated in such a way as to bring the functional side groups to the surface and
inactive side group in the interior.
4.
Find and
write down structures of 10 interesting small molecular bio molecules. Find if
here is any industry which manufactures the components by solution find out who
are the buyers.
Find and
write down structures of 10 interesting small molecular bio molecules. Find if
here is any industry which manufactures the components by solution find out who
are the buyers.
The 10 interesting small molecular bio
molecules are like alcohol, tartaric acid, lactic acid, citric acid, amino
acid, trypsin, sucrose, gluconic acid & amylase.
molecules are like alcohol, tartaric acid, lactic acid, citric acid, amino
acid, trypsin, sucrose, gluconic acid & amylase.
5) Proteins have primary structures, if you are
given a method to know which amino acid is al liker of the two terminal (ends)
of a protein, can you connect this information to purity or homogenising of a
protein ?
given a method to know which amino acid is al liker of the two terminal (ends)
of a protein, can you connect this information to purity or homogenising of a
protein ?

Simply knowing the two amino acids at the
two ends of a protein cannot help in designating a protein as pure or
homogeneous. It is because a protein is made of numerous amino acids. A
difference of only one amino acid any where will make the protein different
from the other. Only by sequencing the complete protein by Edman or songer’s
technique can tell us about purity of a protein.
6. Find out and make a list of protein used as
therapeutic agents Find other applications of proteins
therapeutic agents Find other applications of proteins
THERAPEATIC USES: = Insulin, Oxytocin, ADH,
Thrombin, fibrinogen Renin
Thrombin, fibrinogen Renin
Other
applications –
applications –
a) Cosmetics: Proteins are
used in beauty creams & shampoos e.g. = caesin.
used in beauty creams & shampoos e.g. = caesin.
b) Sweeteners: – Sweetest
chemicals are proteins, threaten followed by monellin.
chemicals are proteins, threaten followed by monellin.
c) Dietary Proteins: It is
added in diet supplements. For building & maintenance of health.
added in diet supplements. For building & maintenance of health.
7. Explain the composition
of triglyceride –
of triglyceride –
They are natural or true fat in a
biochemical formed by esterification of 3 molecules of fatty acids with one
molecule of trihydric alcohol & glycerol. In pure fat, all the 3 fatty
acids of triglyceride are similar (e.g. tripalmitin) while in mixed fat they
are dissimilar (e.g. = dipalmitostearin)
biochemical formed by esterification of 3 molecules of fatty acids with one
molecule of trihydric alcohol & glycerol. In pure fat, all the 3 fatty
acids of triglyceride are similar (e.g. tripalmitin) while in mixed fat they
are dissimilar (e.g. = dipalmitostearin)
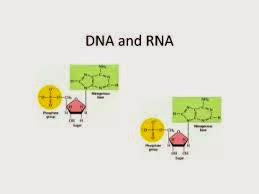
8. Can you describe what
happens when milk is converted into curd or yoghurt from your understanding proteins?
happens when milk is converted into curd or yoghurt from your understanding proteins?
Milk is inoculated with lactic acid bacteria.
They convert milk sugar lactose into lactic acid. Lactic acid acids acts on
globular milk protein casein, causes its denaturation and conversion to fibrous
state. The protein fibres form a reticulum which holds milk fat. This changes
milk into curd or yoghurt. .
They convert milk sugar lactose into lactic acid. Lactic acid acids acts on
globular milk protein casein, causes its denaturation and conversion to fibrous
state. The protein fibres form a reticulum which holds milk fat. This changes
milk into curd or yoghurt. .
9 Can you attempt
building models of bio molecules using commercially available models (ball
& stick models)
building models of bio molecules using commercially available models (ball
& stick models)
Yes, provided balls of different size &
colours are available.
colours are available.
10. Attempt titrating an
amino acid against a weak base & discover the number of dissociating (ionisable/functional
groups in the amino acid.
amino acid against a weak base & discover the number of dissociating (ionisable/functional
groups in the amino acid.
PH of amino acid is recorded. The weak base
is slowly added & the Ph recorded continuously. The number of inflexions
indicate the number of Ionisable functional groups – coat in the acidic range
and – NH2 in the alkaline range.
is slowly added & the Ph recorded continuously. The number of inflexions
indicate the number of Ionisable functional groups – coat in the acidic range
and – NH2 in the alkaline range.
11. Draw the structure of
amino acid alonine
amino acid alonine
CH3
H ______C_______COOH
NH2
12. What are gums made of? Is fevicol different?
Gums are sticky break down products of
plant cell walls consisting of polysaccharides and pectin which exude from the
wounds in the bark. They are generally water soluble.
plant cell walls consisting of polysaccharides and pectin which exude from the
wounds in the bark. They are generally water soluble.
Fevicol is synthetic sticky substance
called resin. Resins are insoluble in water.
called resin. Resins are insoluble in water.
13. Find out a qualitative test for proteins, fats &
oils, amino acids and test any fruit juice saliva, sweat and urine for them.
oils, amino acids and test any fruit juice saliva, sweat and urine for them.
Test
for protein = Biuret test, xanthoproteia test.
for protein = Biuret test, xanthoproteia test.
Test
for fat Oils = Sudan III
for fat Oils = Sudan III
Test
for Amino Acid = ninhydrin test
for Amino Acid = ninhydrin test
14. Find out how much cellulose is made by all the
plants in the biosphere and compares it with how much of paper is manufacture
by man and hence what is the consumption of plant material by man annually.
What is loss of vegetation?
plants in the biosphere and compares it with how much of paper is manufacture
by man and hence what is the consumption of plant material by man annually.
What is loss of vegetation?
About 85 billion tonnes of cellulose is
formed annually in the biosphere (out of 170 billion tones of total organic
matter). Paper making consumes roughly 0.5 billion tonnes of wood food grains
constitute 1.5 billion tonnes. Full wood required is 2 billion tonnes. The
increase in consumption of cellulose has resulted in great loss of vegetation.
formed annually in the biosphere (out of 170 billion tones of total organic
matter). Paper making consumes roughly 0.5 billion tonnes of wood food grains
constitute 1.5 billion tonnes. Full wood required is 2 billion tonnes. The
increase in consumption of cellulose has resulted in great loss of vegetation.
15. Describe the Important properties of enzyme.
1. They are hydrophilic & form hydrosol
in the free state
in the free state
2. They have molecular weight of 6000 to
9,600,000
9,600,000
3. They do not start a chemical reaction
but increase the rate of chemical reaction.
but increase the rate of chemical reaction.
4 .They do not change the
equilibrium but bring out the equilibrium very soon
equilibrium but bring out the equilibrium very soon
If the answers or the article helped
you and you liked the site, please support us to avail you more quality study
materials and solutions .Donate us to support us.
you and you liked the site, please support us to avail you more quality study
materials and solutions .Donate us to support us.